Catalyst Catalyst
Time to settle
Disclaimer / Disclosure long CPRX
December 2, 2025
According to the company,
Catalyst Pharmaceuticals is a commercial-stage biopharmaceutical company focused on acquiring, in-licensing, developing, and commercializing novel medicines for patients living with rare diseases.
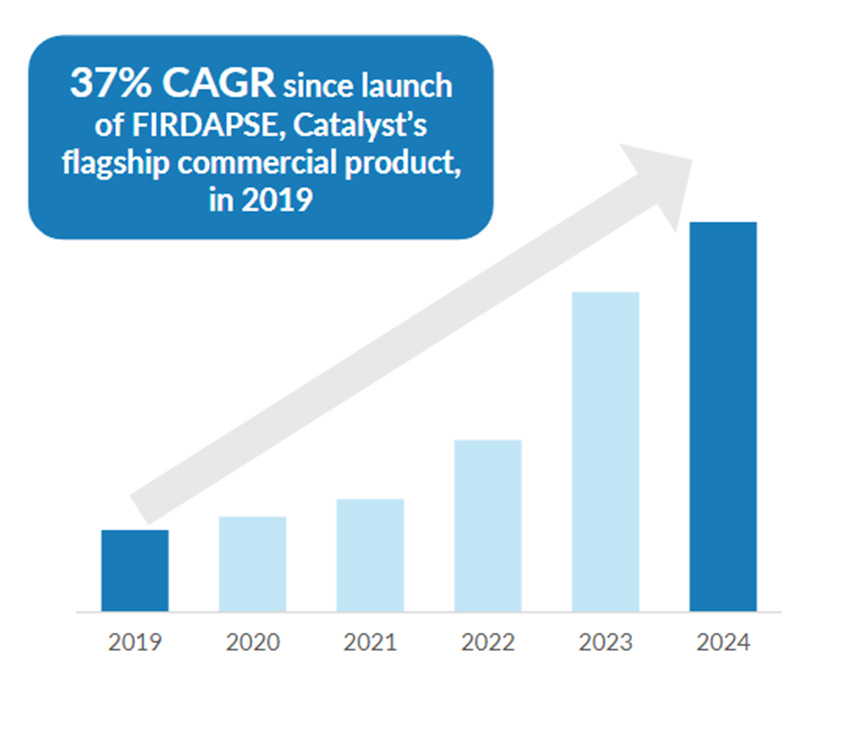
Their Firdapse is the only evidence-based approved product in the U.S. for Lambert-Eaton myasthenic syndrome (LEMS).
Catalyst filed a complaint against Annora earlier this year claiming that Annora’s abbreviated new drug application (ANDA) to market a generic version of Firdapse infringes upon Catalyst’s patents. The trial is scheduled for next quarter. Most of Catalyst’s revenue comes from Firdapse so this is a material issue for the company.
Friday morning, they will hold a settlement conference that could result in an advantageous settlement for Catalyst. So far they settled with both Lupin and Teva on good terms. That leaves the current settlement effort as the last major step in this $2.8 billion market cap company defending their >$1 billion market for the next decade.
Catalyst patents have held up in the face of serious scrutiny from sophisticated adversaries. They are likely to do so once more.
May 7, 2026
Angelini Pharma to Acquire Catalyst for $31.50 per share
The Boards of Directors of Angelini Pharma and Catalyst Pharmaceuticals have unanimously approved the acquisition of Catalyst Pharmaceuticals at 31.50 USD per share in cash, for a total consideration of approximately 4.1 billion USD, representing a premium of 28% to the 30-day volume-weighted average trading price as of April 22, 2026
The transaction marks Angelini Pharma’s entry into the U.S. market, reinforcing its long-term commitment to Brain Health and its dedication to people living with Rare Diseases
Closing is expected in the third quarter of 2026
ROME and CORAL GABLES, Fla., May 07, 2026 (GLOBE NEWSWIRE) -- Angelini Pharma S.p.A. (“Angelini Pharma”), an international pharmaceutical company and part of the Angelini Industries Group, and Catalyst Pharmaceuticals, Inc. (”Catalyst”) (Nasdaq: CPRX), a commercial-stage biopharmaceutical company focused on in-licensing, developing, and commercializing novel medicines for patients living with rare and difficult-to-treat diseases, today announced that they have entered into a definitive agreement pursuant to which Angelini Pharma has agreed to acquire all outstanding shares of Catalyst for 31.50 USD per share in cash, for a total equity value of approximately 4.1 billion USD (equivalent to 3.5 billion euros) which represents a 21% premium to Catalyst’s unaffected closing share price on April 22, 2026, the last trading day before market signs that the transaction had become public information, as well as a 28% premium to the 30-day volume-weighted average trading price to that unaffected date. The transaction has been unanimously approved by the Boards of Directors of both companies and is expected to close in the third quarter of 2026.
This acquisition marks a pivotal moment in the Angelini Group’s history — a group with over 100 years of history, chaired by Thea Paola Angelini, a fourth-generation member of the Angelini family — led by Sergio Marullo di Condojanni, Chief Executive Officer of Angelini Pharma.
The transaction will be carried out with the participation of Blackstone funds and select international partners, and will be financed with the support of BNP Paribas, acting as Sole Global Coordinator and Underwriter of the financing package.
Founded in 2002 and listed on Nasdaq since 2006, Catalyst has built a robust portfolio of products focused on the treatment of rare neuromuscular and neurological diseases: FIRDAPSE® (amifampridine), the only evidence-based treatment approved by the U.S. Food and Drug Administration (FDA) for Lambert-Eaton myasthenic syndrome (LEMS) in patients aged six and above; AGAMREE® (vamorolone) a novel corticosteroid approved by the FDA in 2023 for the treatment of Duchenne Muscular Dystrophy (DMD) in patients aged 2 and above; and FYCOMPA® (perampanel), an antiepileptic drug approved for the treatment of partial-onset seizures and primary generalised tonic-clonic seizures, the U.S. rights to which were acquired from Eisai in 2023.
Following completion of the acquisition, Angelini Pharma intends to integrate Catalyst’s portfolio and exceptional commercial infrastructure with its own expertise and products in Brain Health to develop a next-generation therapeutic platform in Rare Diseases. This transaction reinforces the commitment Angelini Pharma has built over recent years through products such as Ontozry® and high-profile scientific collaborations. Catalyst’s portfolio is expected to significantly strengthen the company’s strategic objectives, growing its presence in the U.S. as part of a balanced strategy that aims to develop the North American market while continuing to strengthen its core business in Europe. Angelini Pharma’s continued industrial presence in Italy will remain a strategic asset as a valuable production and scientific center within the combined company’s global operations.
Transaction Terms
Under the terms of the agreement, a subsidiary of Angelini Pharma will merge with and into Catalyst, and, following the merger, Catalyst will survive as a wholly owned subsidiary of Angelini Pharma. Angelini Pharma expects to finance the acquisition with a combination of cash and debt. The acquisition is not subject to any financing condition.
The agreement has been unanimously approved by the Boards of Directors of both companies. The parties expect to close the acquisition in the third quarter of 2026, subject to approval by Catalyst stockholders, the receipt of required regulatory approvals, and satisfaction of other customary conditions. Due to the pendency of the transaction, Catalyst’s 2026 annual meeting of stockholders will be suspended.
Pending U.S. FIRDAPSE IP Litigation Resolved
Separately, Catalyst announced today that it has resolved the patent litigation it brought in response to an Abbreviated New Drug Application filed on behalf of Hetero USA, Inc, (together with its affiliated parties who are parties to the litigation, “Hetero”) seeking approval to market a generic version of FIRDAPSE® (amifampridine)10 mg tablets prior to expiration of the applicable Catalyst patents. In connection with the resolution of the patent litigation, Catalyst and its licensor SERB S.A. have entered into a Settlement Agreement with Hetero. As required by law, the companies will submit the confidential settlement agreement to the U.S. Federal Trade Commission and the U.S. Department of Justice for review. In accordance with the agreement, the parties will terminate all ongoing patent litigation between Catalyst/SERB and Hetero regarding FIRDAPSE patents pending in the U.S. District Court for the District of New Jersey. This settlement resolves all pending patent litigation relating to FIRDAPSE.
Five years ago, we embarked on a profound transformation of Angelini Pharma — organizational, scientific and strategic — with the ambition to build a company capable of competing at the highest global level. On one hand, we continued to invest in our traditional portfolio; on the other, we chose to focus on central nervous system disorders, with the goal of addressing an unmet medical need that is, unfortunately, growing significantly. We invested in innovation through the development of a high-value asset pipeline, including collaborations with leading partners such as Blackstone Life Sciences in GRIN Therapeutics. Today, we take another significant step with the acquisition of Catalyst Pharmaceuticals, which we believe will establish Angelini Pharma as a relevant global player in neurological Rare Diseases. Entering the U.S. market will allow us to acquire the scale and capabilities needed to continue this journey. Patient care remains at the heart of our vision. We continue to look ahead with determination, backed by a clear strategy and the drive to keep growing on a global scale. We are proud of a transaction that demonstrates, once again, the dynamism of the Italian pharmaceutical industry.
— Sergio Marullo di Condojanni, CEO of Angelini Pharma
This is a pivotal and transformative moment for Catalyst, our team, and the patients we serve. By combining our unique capabilities in rare diseases with Angelini’s proven global reach, we will create a stronger, scalable, and robust rare disease platform to expand access to life-changing therapies worldwide. For shareholders, this transaction delivers immediate and certain cash value through a compelling premium. We are proud of the incredible foundation our team has built and are confident that together with Angelini, we can enhance patient support, accelerate innovation, and continue to drive sustainable long-term value for all stakeholders.
— Rich Daly, President and CEO of Catalyst
Assuming that the deal closes on current terms by October, the $0.31 net spread offers a 3% prospective IRR which is pretty meh unless other bidders emerge.